Soils Glossary
Select an item from the list above to see the definition. Hitting a letter on your keyboard will go straight to that point in the list.
A HORIZON
Mineral horizons close to the soil surface which are darkened due to organic matter accumulation, and/or appear to have lost clay, iron or aluminium with resultant concentration of quartz or other resistant minerals.
A1 HORIZON
An A horizon at the soil surface which is characterised by relatively high organic matter and biological activity and a dark colour compared to horizons below.
A2 HORIZON
An A horizon which is paler than A1 horizon above and a B horizon or other soil material below.
ACCELERATED EROSION
Water or wind erosion resulting from human activity which is more rapid than normal or geological rates.
ACTINOMYCETES
A group of filamentous bacteria, which may be abundant in soils.
ADSORPTION
The process whereby ions or molecules are attracted and held close to the surfaces of soil minerals. For example cations are adsorbed by clay minerals carrying negative charges.
AEOLIAN
Pertaining to sediments transported and deposited by wind. Parna, loess and sand dunes are examples.
AEROBIC
An aerobic soil environment is one where there is free oxygen (ie O2) present. The opposite of anaerobic. See also respiration.
AGGREGATE
A soil particle formed when primary soil particles (sand, silt and clay) are bonded together. Peds are examples of soil aggregates. Contrast with primary particle.
AGGREGATE STABILITY
The ability of compound particles of soil to maintain integrity when a disruptive force is applied.
AIR-DRY
Soil samples are air-dry when they have been allowed to dry at room temperature to a stable water content. The remaining water can be removed by heat to produce oven-dry soil.
AIR-FILLED POROSITY
The proportion of the total soil volume that is occupied by air.
ALLOPHANE
A noncrystalline aluminosilicate soil clay mineral.
ALLUVIUM
Material deposited by flowing water.
ALPINE HUMUS
A great soil group. An alpine humus is a strongly acid organic soil with a thick loamy A horizon overlying a coarser textured C horizon. They form on a variety of parent materials and are restricted to the highlands of south-eastern Australia. The Great Soil Group system is now superseded by the Australian Soil Classification.
ALUMINOSILICATE
Minerals containing aluminium, silicon and oxygen as the main elemental constituents..
AMMONIA VOLATILISATION
The loss of ammonia, and hence nitrogen, from the soil as ammonia gas escapes to the atmosphere.
AMORPHOUS
Amorphous minerals lack a regular crystal structure.
AMPHIBOLES
A group of primary aluminosilicate minerals with a crystal structure comprising double chains of linked silica tetrahedra. Hornblende is a common example.
ANAEROBIC
An anaerobic soil environment lacks free (molecular) oxygen (eg a waterlogged soil). The opposite of aerobic. See also respiration.
ANGULAR BLOCKY
Angular blocky peds are square or block like with relatively sharp and well defined edges and vertices
ANION EXCHANGE CAPACITY
The number of moles of charge of anions that are adsorbed exchangeably per unit mass of soil.
ANTHROPOSOL
A soil order in the Australian Soil Classification. Anthroposols are soils resulting from human activities
APEDAL
Soil structure is apedal if it lacks peds. Apedal structures may be massive or single grain. Contrast with pedal.
AQUIFER
A porous layer within the soil or underlying in which water is stored.
ARABLE
Land which can be cultivated for the production of crops.
ASPECT
The aspect of a slope is the direction you are facing if you are looking down the slope.
ATTERBERG LIMITS
The soil plastic and liquid limits. Used in soil engineering to characterise how soil consistence varies with water content.
AUSTRALIAN SOIL CLASSIFICATION
Developed by R.F. Isbell and introduced in 1996, the Australian Soil Classification is a hierarchical scheme which uses field and laboratory data to classify soil profiles via a key. The levels in the hierarchy are: order, suborder, great group, subgroup and family. The soil is assigned a name which suggests some of the properties of the soil. There are 14 soil orders: Anthroposols, Calcarosols, Chromosols, Dermosols, Ferrosols, Hydrosols, Kandosols, Kurosols, Organosols, Podosols, Rudosols, Sodosols, Tenosols, and Vertosols.
B HORIZON
A horizon which shows signs of illuviation (accumulation of clay, iron, aluminium, or organic matter), or differs from the A horizons above or horizons below in structure, consistence or by being more strongly coloured.
BACTERIA
A large group of procaryotic microorganisms which are abundant in soil and play a major role in nutrient and organic matter cycling.
BASE SATURATION
The proportion of the cation exchange capacity that is occupied by the exchangeable bases, expressed as a percentage.
BED LOAD
Large, heavy particles that are moved by water (eg in streams) by creep.
BIOTITE
A dark coloured mica common in some granites.
BIOTURBATION
A form of pedoturbation (soil mixing) caused by living organisms (eg burrowing by worms, ants, termites, etc, or tree throw caused by the roots of falling trees).
BLACK EARTH
A great soil group which is dark coloured, has a uniform clay texture profile, cracks upon drying and contains calcium carbonate at depth. The Great Soil Group system is now superseded by the Australian Soil Classification.
BROWN CLAY
A member of the grey, brown and red clays great soil group. Deep, uniform cracking clay soils widespread in the alluvial plains of inland Australia. The Great Soil Group system is now superseded by the Australian Soil Classification.
BUFFER CAPACITY
A measure of the ability of the soil to resist change in pH when acid or alkali is added. May also be applied to other chemical species. For example the P buffer capacity of a soil is the degree to which it resists change in soil solution P concentration when soluble P is added or removed.
BULK DENSITY
The mass of oven-dry soil divided by the bulk soil volume at sampling. Contrast with particle density.
C HORIZON
Layers below the solum (A and B horizons) which have usually been partly weathered, but are little affected by soil forming processes such as pedoturbation. Often the structure of the underlying parent rock is still visible.
C/N RATIO
The ratio by mass of carbon to nitrogen in organic matter.
CALCAROSOL
A soil order in the Australian Soil Classification. Calcarosols are characterised by pedogenic carbonate material through much or all of the solum.
CAPILLARY FORCES
Forces of attraction between water and the soil surface which help retain water in soil micropores.
CARBOHYDRATE
A group of organic compounds composed of carbon, hydrogen and oxygen. Examples are sugars like glucose and sucrose, and polysaccharides like starch and cellulose.
CARBOXYL
The organic functional group -COOH. Along with phenol groups, carboxyl groups are responsible for the acidity and cation exchange capacity of humus.
CATION EXCHANGE CAPACITY
The amount of cations that a soil can adsorb exchangeably, expressed as moles of charge per unit soil mass. Equals the amount of negative charge possessed by the soil colloids.
CEC
See cation exchange capacity.
CELLULOSE
A carbohydrate which is abundant in plants and plant residues in soil.
CHELATE
A chemical compound in which a molecule (in soils usually an organic acid) wraps around and bonds to a polyvalent metal cation (eg ferric or aluminium ions). Chelation can increase the solubility of ions like aluminium and iron in soils.
CHOCOLATE SOIL
A great soil group. A brown, well aggregated soil commonly formed in situ from basalt in an upper slope position. The Great Soil Group system is now superseded by the Australian Soil Classification.
CHROMA
The intensity or saturation of a soil colour. See also Munsell colour.
CHROMOSOL
A soil order in the Australian Soil Classification. Chromosols have a strong texture contrast between the A and B horizons, and the B horizons are neither sodic nor strongly acidic.
CHRONOSEQUENCE
A series of soils for which all the factors of soil formation apart from time are approximately constant.
CLAY
In the International system (used in Australia), clay is defined as soil particles less than 0.002 mm in diameter.
CLAY MINERALS
Secondary minerals which commonly occur in the clay fraction of soils and include layer silicate minerals such as kaolinite, montmorillonite and illite, as well as non-silicate minerals such as hematite, goethite and gibbsite. 1:1 clay minerals are silicate clay minerals with one tetrahedral layer to one octahedral layer (eg kaolinite). 2:1 clay minerals are silicate clay minerals with two tetrahedral layers to one octahedral layer (eg illite and montmorillonite).
CLOD
A soil aggregate produced by artificial means such as by ploughing or digging. In contrast peds are naturally occurring soil aggregates.
COHERENCE
The degree to which soil material is bound together. Soil is said to be coherent if more than two thirds of the material (whether in peds or not) is united at a given soil water content and in the absence of an applied force. For example, a massive clay is coherent, but beach sand lacks any coherence. Synonymous with cohesiveness.
COHESIVENESS
See coherence.
COLLOID
Material composed of particles less than 0.002 mm in diameter.
COLLUVIUM
Material that has crept downslope under the force of gravity and has accumulated at the foot of the hill.
COLOUR
See Munsell colour.
COLUMNAR
A type of soil structure where peds have a vertical axis that is much longer than the horizontal axis and have domed tops. Compare with prismatic structure.
COMPACTION
The process of compression of soil into a smaller volume. Bulk density is a measure of the result of a compaction process. As a soil becomes more compacted, its bulk density increases.
CONSERVATION FARMING
Farming that emphasises conservation of soil and water resources. Includes practices such as minimising tillage and retaining crop residues.
CONSISTENCE
Field characterisation of the behaviour of soil when it is subject to stress. Consistence varies with water content, and includes assessment of strength, stickiness and plasticity. See also Atterberg limits.
CONTOUR BANK
An erosion control measure where soil has been pushed up into a bank nearly on the contour, so that runoff is caught and redirected to a non-erodible slope such as a grassed waterway.
CRACKING CLAYS
A general term (not part of any soil classification scheme) to describe uniform clay soils which shrink to form a pattern of deep cracks upon drying.
CRUST
A surface crust is a thin hard layer formed at the soil surface as a result of structural collapse associated with raindrop impact.
CUTAN
A thin layer on the outside of a ped which is different to the soil material within the ped. Also called a clay skin. Cutans often have a polished appearance and are of a different colour to the interior of the ped. In clay B horizons they may be produced by clay illuviation.
CYCLIC SALT
Salts derived from the sea that are carried inland as aerosols and deposited in the landscape.
DEEP DRAINAGE
Drainage of water through the soil profile below the root zone.
DEFLOCCULATION
See dispersion.
DENITRIFICATION
The reduction of nitrate in soil by microorganisms to produce nitrous oxide (N2O) and dinitrogen (N2) gases.
DERMOSOL
A soil order in the Australian Soil Classification. Dermosols have pedal B horizons but lack strong texture contrast between the A and B horizons.
DESORPTION
Release of an adsorbed ion or molecule from a surface. The opposite of adsorption.
DIFFUSION
Movement of molecules or ions down a concentration gradient (ie from a place of high concentration to one of low concentration).
DIRECT DRILL
Sowing seeds directly into the soil without prior cultivation. This term is mainly used in southern and western Australia. See also no-till and fallow.
DISPERSION
The process whereby the clay fraction of soil aggregates is broken down into primary particles. Dispersion in water can be recognised by an increase in the turbidity of the water as clay particles form a colloidal suspension. Synonymous with deflocculation. See also slaking and flocculation.
DUPLEX
A duplex soil is one which has a duplex texture profile, which means there is an abrupt and marked increase in clay content with depth.
E HORIZON
An A2 horizon. Not used in Australia.
ELUVIATION
The soil forming process whereby soil material is moved out of a soil layer in suspension.
ERODIBILITY
The susceptibility of soil to erosion
EROSION
The process whereby soil is removed by the action of water or wind.
EROSIVITY
The strength of the tendency of water (rain or overland flow) to cause erosion.
ESP
The ESP or exchangeable sodium percentage is the percentage of the effective cation exchange capacity of the soil occupied by sodium ions.
EVAPORATION
Loss of water as vapour to the atmosphere directly from the soil. Contrast with evapotranspiration and transpiration.
EVAPOTRANSPIRATION
The sum of evaporation and transpiration.
EXCHANGE ACIDITY
The sum of exchangeable aluminium and hydrogen ions in the soil.
EXCHANGEABLE BASE
An exchangeable cation other than hydrogen or aluminium ions. The most abundant exchangeable bases in soils are sodium, potassium, calcium and magnesium ions. Note that exchangeable bases are not bases in the strict chemical sense (ie proton acceptors). However soils that have high (close to 100%) base saturation do tend to be alkaline or neutral, because of the absence of the truly acidic cations of aluminium and hydrogen. Contrast with exchange acidity.
EXCHANGEABLE IONS
Cations or anions that are held by electrostatic attraction to an oppositely charged surface (eg clay particle) and which may be displaced by other ions.
EXCHANGEABLE SODIUM PERCENTAGE
The ESP or exchangeable sodium percentage is the percentage of the effective cation exchange capacity of the soil occupied by sodium ions.
EXTERNAL DRAINAGE
Drainage of water away from a site by runoff rather than through the soil profile. Contrast with internal drainage.
FABRIC
The appearance of the surface of soil material. In the field a hand lens is often used for close inspection. Where few, if any, peds are present the fabric is described as sandy (like beach sand) or earthy (a matt appearance due to the presence of fine pores). If peds are evident, the fabric is described as smooth-ped (peds dense and smooth-faced) or rough-ped (peds have relatively porous surfaces and are rough-faced).
FACTORS OF SOIL FORMATION
The environmental factors which define the nature of the soil formed at a particular place in the landscape. Five are usually identified: parent material, climate, topography, biota and time.
FACTUAL KEY
See Northcote Key.
FALLOW
A period in the cropping cycle where the land is kept free of growing plants. This allows soil water and mineralised nutrients to accumulate. Weeds need to be controlled during the fallow, and this is achieved by cultivation or herbicides. The plant residues from the previous crop have traditionally been removed by burning (a bare fallow), but are increasingly being retained. Retained residues may be left untouched (no-till fallow), incorporated into the soil by tillage (stubble incorporation), or chopped and left on the soil surface (stubble mulching).
FELDSPAR
The most abundant group of primary aluminosilicate minerals, being found in all the common igneous rocks. Plagioclase feldspars contain Ca and Na, and alkali feldspars contain K and Na.
FERROSOL
A soil order in the Australian Soil Classification. Ferrosols lack strong texture contrast between the A and B horizons, and have B horizons which are high in free iron oxide.
FIELD CAPACITY
The amount of water retained in the soil after it has been saturated and allowed to drain freely for 1 to 2 days. Often approximated by the soil water content at a matric potential of -10 kPa.
FIELD TEXTURE
Texture as determined by a field method, such as the ribbon test. Is influenced by factors in addition to particle size distribution.
FINE EARTH
The fraction of soil particle sizes with diameters less than 2 mm (ie clay + silt + sand). Soil chemical and physical analyses are usually carried out on the fine earth fraction after the gravel (> 2 mm)has been removed by sieving.
FLOCCULATION
The bonding together of soil colloids (clay particles) that are suspended in water to form compound particles (microaggregates). When a colloidal suspension flocculates gravity causes the particles to rapidly settle out. Flocculation is the opposite of deflocculation or dispersion.
FLUX
The amount of a quantity transported per unit time. For example in soil hydrology the infiltration flux is the rate at which water enters the soil. In nutrient cycling nutrient flux is the rate at which a nutrient moves from one pool to another.
FRIABLE
Easily crumbled. For example a loam with a high organic matter level would be friable when moist.
FULVIC ACID
Complex organic molecules that form part of soil humus and are soluble in water.
FUNGAL HYPHAE
The individual filaments which make up the body of a fungus.
GEOLOGICAL EROSION
Erosion occurring at natural rates, and hence a natural soil and landscape forming process. Contrast with accelerated erosion.
GIBBSITE
A white sesquioxide soil clay mineral with formula Al(OH)3.
GILGAI
A form of soil surface microrelief comprising hummocks and/or hollows. Associated with the swelling and shrinking that occurs in expansive soils as they go through wetting and drying cycles. Linear gilgai are long parallel mounds and depressions that run down a gentle hill slope (relief usually less than 300 mm, mounds usually separated by 5 to 8 m). Melonhole gilgai are irregular large depressions on level ground usually more than 3 m in diameter and more than 300 mm deep.
GLEY
Dull grey or bluish-grey colours produced in soil by extended periods of waterlogging and the chemical reduction or removal of iron minerals.
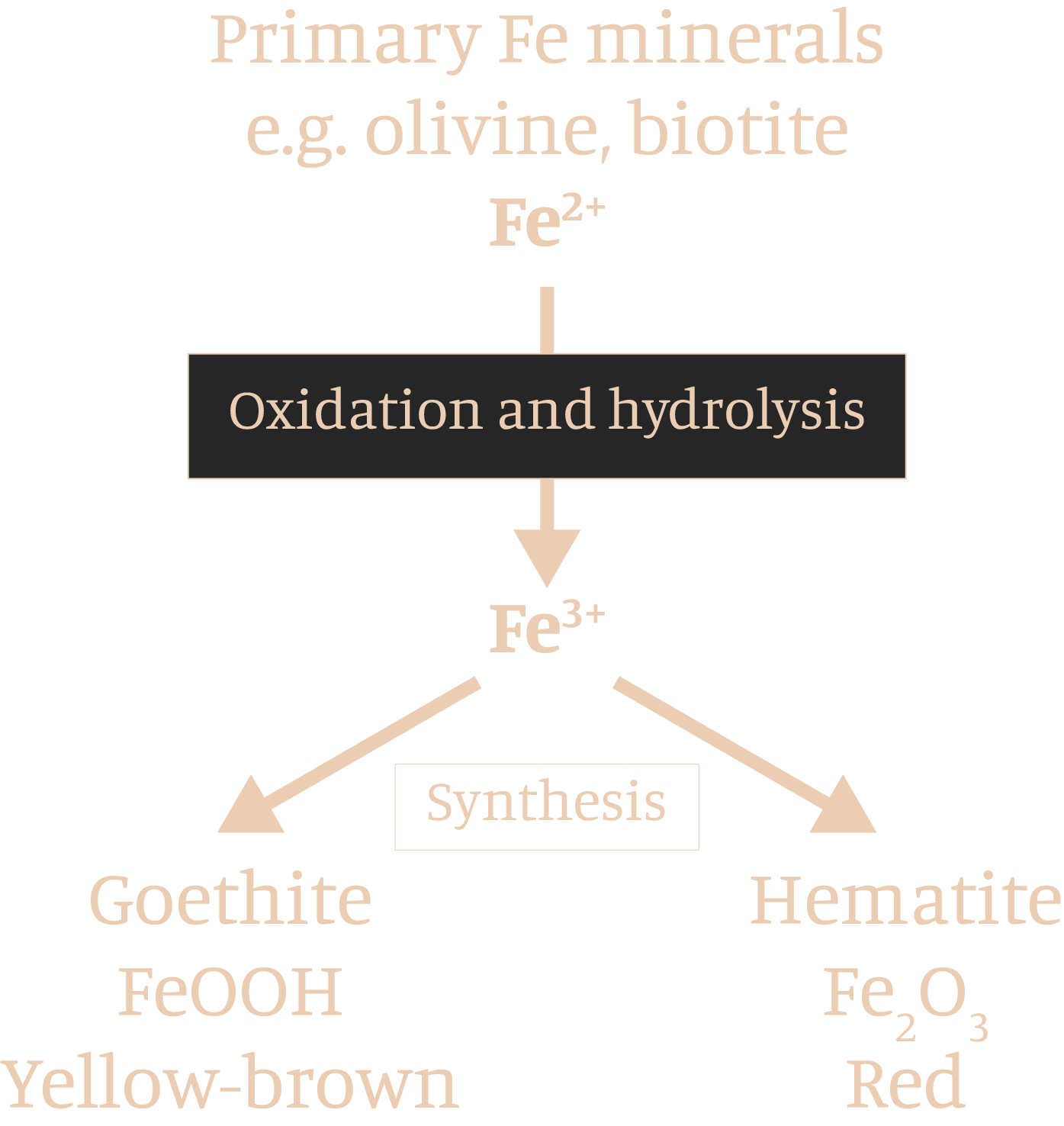
GOETHITE
A sesquioxide soil clay mineral with formula FeOOH, and which is commonly responsible for yellow-brown colours in soils.
GRADATIONA
A gradational soil is one which has a gradational texture profile, which means that the texture gradually becomes heavier (more clayey) with depth.
GRANULAR
A structure with spheroidal peds that do not fit into the faces of adjacent peds.
GRASSED WATERWAY
A strip of land running down a slope which is kept permanently covered with grass and used as a safe channel to transport water down the slope without causing erosion. Often used in conjunction with contour banks.
GRAVEL
In the International scheme (used in Australia), gravel is the fraction of soil particles with diameter greater than 2 mm. Contrast with fine earth.
GRAVIMETRIC WATER CONTENT
The mass of water contained in a soil sample divided by the mass of oven-dry soil. Also termed mass water content.
GRAVITATIONAL POTENTIAL
The component of water potential that is caused by the position of the water in the earth#39;s gravitational field (ie its elevation). Gravitational potential is measured relative to some arbitrary reference height (eg the soil surface) and is positive above that point and negative below that point. See also water potential.
GREAT SOIL GROUP
One of 43 Australian soil types described in "A Handbook of Australian Soils". The Great Soil Group system is now superseded by the Australian Soil Classification.
GREEN MANURE
A crop grown and then ploughed into the soil without harvesting in order to improve soil fertility.
GREY CLAY
A member of the grey, brown and red clays great soil group. Deep, uniform cracking clay soils widespread in the alluvial plains of inland Australia. The Great Soil Group system is now superseded by the Australian Soil Classification.
GULLY
An erosion channel cut into the soil by a concentration of runoff. Gullies are deeper than rills and are not able to be ploughed out. See also rill and sheet erosion.
GYPSUM
The mineral name for calcium sulfate dihydrate.
HARDSETTING
The soil degradation process whereby a surface soil horizon dries to a hard apedal mass.
HEMATITE
A sesquioxide mineral with formula Fe2O3, that is often found in the clay fraction of soils, and which is responsible for the red colours of soils.
HORIZON
A soil layer which is approximately parallel to the land surface and differs in properties from adjacent layers.
HUE
The component of soil colour which indicates its dominant spectral wavelength (eg yellow or red). See also Munsell colour.
HUMIC ACID
A component of soil humus which is insoluble in water but soluble in dilute alkali.
HUMIN
A component of soil humus which is insoluble in water and also insoluble in dilute alkali.
HUMUS
The relatively stable, dark coloured mixture of organic compounds which is produced as plant or animal residues decompose in the soil.
HYDRAULIC CONDUCTIVITY
A measure of the ability of the soil to conduct water. More precisely it is the flux of water at unit hydraulic potential gradient. The saturated hydraulic conductivity is the hydraulic conductivity when the soil is saturated.
HYDRAULIC POTENTIAL
The sum of the matric, pressure and gravitational components of water potential. Differences in hydraulic potential determine the movement of water within the soil profile. Movement of water into plant roots, however, is also influenced by differences in solute (osmotic) potential. See also water potential.
HYDROSOL
A soil order in the Australian Soil Classification. Hydrosols include a range of soils which are seasonally or permanently wet.
IGNEOUS
Igneous rocks are those formed as magma cools and solidifies.
ILLITE
A 2:1 clay mineral related in structure and composition to mica, and in which the layers are held together by unhydrated potassium ions.
ILLUVIATION
The soil forming process whereby soil material in suspension is transported into and accumulates in a soil layer.
IMMOBILISATION
Also spelled immobilization. The process whereby inorganic chemical forms of an element are converted to organic forms. For example, if a microorganism takes up nitrate and converts the N to protein, immobilisation of N has occurred. The opposite of mineralisation.
INFILTRATION
Movement of water through the soil surface and into the soil.
INTERNAL DRAINAGE
Drainage of water through the soil profile. Contrast with external drainage.
IRONSTONE
Hardened material often found in soil as concretions and which is rich in iron oxides, but also often containing substantial quantities of aluminium and manganese oxides. See also laterite.
ISOMORPHOUS SUBSTITUTION
Substitution of one ion in a crystal structure with another without altering the structure. In clay minerals isomorphous substitution of one cation with another of a lower charge gives the structure a permanent negative charge which contributes to its cation exchange capacity.
KANDITE
A group of 1:1 clay minerals clay. Kaolinite is a common example.
KANDOSOL
A soil order in the Australian Soil Classification. Kandosols have weakly pedal or massive B horizons, little texture contrast, and are not calcareous throughout.
KAOLINITE
A 1:1 clay mineral common in soils.
KRASNOZEM
A great soil group which is typically red, high in clay content, leached and acid. The Great Soil Group system is now superseded by the Australian Soil Classification.
KUROSOL
A soil order in the Australian Soil Classification. Kurosols have a strong texture contrast between the A and B horizons and the B horizons are strongly acid
LAND CAPABILITY
The ability of land to sustainably support a given land use.
LATERITE
A zone of ironstone within the soil which may form a continuous mass or be composed of concretions.
LAYER SILICATES
Those aluminosilicate minerals which have crystal structures comprising octahedral and tetrahedral layers, yielding plate like crystals. The silicate clay minerals and micas are layer silicates.
LEACHING
The removal from the soil of materials dissolved in percolating water. Contrast with eluviation
LENTICULAR
Peds which are lens shaped.
LIGNIN
Lignin is a complex polymer of phenolic compounds which gives strength to woody tissue. In soil lignin is quite resistant to decay, and partially altered lignin is a component of humus.
LIQUID LIMIT
The water content at which a soil passes from a plastic (retains its shape after being deformed) to a liquid state (flows).
LITHOSOL
A great soil group which includes soils that are shallow and stony. The Great Soil Group system is now superseded by the Australian Soil Classification.
LOAM
A mid range soil texture class containing approximately equal quantities of clay, silt, fine sand and coarse sand.
LOESS
Soil material predominantly of silt (with some clay) size that has been deposited after being transported by wind. Also see parna.
MACRONUTRIENTS
The plant nutrients that are needed in the largest quantities: N, P, K, S, Ca and Mg.
MACROPORES
Soil pores greater than 0.5 mm in diameter.
MASS WATER CONTENT
See gravimetric water content.
MASSIVE
Soil structure is massive if no peds are evident (ie it is apedal) and the soil is coherent (soil primary particles are bonded together).
MATRIC POTENTIAL
The component of water potential that results from the adsorption of water molecules onto soil particle surfaces and the capillary attraction of soil pores for water. In unsaturated soil the matric potential is negative, reaching zero at saturation. Pressure potential and matric potential are sometimes lumped together, as pressure potential is zero in unsaturated soil, but positive in saturated soil. See also water potential.
MICA
A group of primary aluminosilicate minerals with a layered crystal structure similar to that of the 2:1 silicate clay minerals. Two common forms are biotite and muscovite.
MICRONUTRIENTS
Plant nutrients that are required in relatively small quantities (though they are still essential for plant growth). Micronutrients include: Cl, B, Zn, Fe, Mo, Mn, Cu and Ni.
MICROPORES
Pores less than 0.5 mm in diameter.
MINERALISATION
Also spelled mineralization. The process whereby organic chemical forms of an element are converted to inorganic forms. For example, if, as humus is slowly decomposed, ammonium is released into the soil, then mineralisation of N has taken place. The opposite of immobilisation.
MINERALS
Soil minerals are the solid inorganic constituents of soil, each of which has defined chemical and crystalline properties. Primary minerals (eg quartz) were formed as igneous rocks crystallised and have been inherited chemically unaltered by the soil. Secondary minerals (eg clays) have been formed by the chemical weathering of primary minerals.
MINIMUM TILLAGE
Part of conservation farming practice in which the number of tillage operations is reduced to the minimum necessary for crop production.
MONTMORILLONITE
A member of the smectite group of 2:1 clay minerals. Sometimes montmorillonite is used synonymously with smectite.
MOTTLES
Patches in a soil horizon which have a different colour to the dominant colour for that horizon.
MULCH
Material which is spread over the surface of the soil, and which modifies conditions near the soil surface (eg by altering soil temperature, evaporation or erodibility).
MUNSELL COLOUR
The colour of soil described objectively in terms of hue, value and chroma by reference to the Munsell colour charts.
MUSCOVITE
A pale coloured mica common in some granites.
NITRIFICATION
The oxidation of ammonium to nitrate by microorganisms in soil.
NITROGEN FIXATION
Conversion of atmospheric dinitrogen gas (N2) to a chemical form than plants can use (eg ammonium or nitrate).
NO-TILL
A cropping system where there is no tillage, the stubble is untouched during the fallow, weeds are controlled with herbicide, and the next crop is sown directly in the standing stubble. In Australia this term is mainly used in northern New South Wales. See also direct drill and fallow.
NORTHCOTE KEY
A key developed by Keith Northcote in the 1960s and formerly used for classifying soils in Australia based on objective assessment of soil profiles. Soils were given a letter and number code (eg Dr4.21). Also termed the Factual Key.
NUTRIENT
In general, a nutrient is a substance which contributes to the growth and functioning of an organism. In the context of soils, the term nutrient is often taken as synonymous with plant nutrient. The essential plant nutrients are those elements (apart from C, H and O) which are essential for plants to complete their life cycle. Currently the nutrients which are known to be essential to plants in general are: N, P, K, S, Ca, Mg, Cl, B, Zn, Fe, Mo, Mn, Cu and Ni.
NUTRIENT CYCLE
A nutrient cycle is the set of pools and fluxes that describe the translocation and transformation of a nutrient through an ecosystem.
OCTAHEDRON
An eight sided volume with 6 vertices. In soil clay minerals an aluminium ion is often surrounded by 6 oxygen anions situated at the vertices of an octahedron. Linked octahedrons form the octahedral layers in clay mineral crystals.
ORGANIC MATTER
Soil organic matter comprises a wide range of organic carbon compounds which were originally part of living organisms.
ORGANIC SOIL
A soil with a high proportion of organic matter throughout the solum.
ORGANOSOL
A soil order in the Australian Soil Classification. Organosols are dominated by organic materials.
OSMOTIC POTENTIAL
See solute potential.
OVEN-DRY
Soil samples are oven-dry when they have been allowed to dry in an oven at 105 C to a stable water content. Usually this takes about 24 hours.
PARENT MATERIAL
The material from which the soil is formed.
PARNA
Aeolian deposits of calcareous clay which are common in the Riverina.
PARTICLE DENSITY
The density of the particles within the soil, often assumed to be 2.65 grams per cubic centimetre. Contrast with bulk density.
PEAT
An organic soil formed where organic matter decomposition has been impeded by permanent waterlogging.
PED
Peds are aggregates that occur naturally in the soil profile. They are separated one from another in the soil profile by planes of weakness which may be indicated by cracks or cutans. Peds are different to clods, which are formed artificially (eg by tillage).
PEDAL
Soil structure is pedal if peds are present. Contrast with apedal.
PEDOGENESIS
Soil formation. Hence pedogenic means formed within the soil profile.
PEDOLOGY
The study of soils in relation to their genesis, classification and distribution over the landscape.
PEDOTURBATION
A soil forming process in which soil material is mixed within the soil profile. Examples of pedoturbation are the shrinking and swelling of cracking clays leading to gilgai, or bioturbation due to burrowing animals.
PENETRABILITY
The ease with which a soil can be penetrated. See also penetrometer.
PENETROMETER
A device consisting of a steel rod with a conical tip that is used to measure soil strength.
PERCOLATION
The downward movement of water though the soil under the influence of gravity.
PERMANENT CHARGE
That component of the electrical charge possessed by soil colloids which does not vary with environmental conditions. Permanent charge is related to isomorphous substitution in the clay crystal lattice. Contrast with variable charge.
PERMANENT WILTING POINT
The water content of soils at which plants will not regain turgor even if the soil water content is raised. Often taken as the lower limit at which plants can use soil water and approximated by the soil water content at a matric potential of -1500 kPa.
PHASE RELATIONS
The relationship between the solid, liquid and gaseous components of soil.
PHOSPHORUS FIXATION
The process by which phosphate ions become adsorbed so strongly by soil colloids that they are no longer exchangeable.
PLANT AVAILABLE WATER
Soil water that plants are able to take up and transpire. Excludes soil water held in large pores that drain rapidly and soil water held in pores so fine that the plant roots cannot extract it. Numerically plant available water is equated to the difference in water content between field capacity and permanent wilting point.
PLASTIC LIMIT
The water content at which a soil passes from a solid to a plastic state. Operationally defined as the water content at which soil can be rolled into 3 mm diameter rods without the rods breaking. Tillage when the soil is at or wetter than the plastic limit is likely to result in compaction or smearing. The plasticity index is the numerical difference between the liquid limit and the plastic limit. See also Atterberg limits.
PLATY
A type of soil structure where the horizontal dimensions of peds exceed the vertical dimension.
PODOSOL
A soil order in the Australian Soil Classification. Podosols have B horizons which characterised by accumulations of organic matter, iron and/or aluminium.
PODZOL
A great soil group characterised by a uniform sandy profile where a B horizon has been formed by the illuviation of humus and sesquioxides. Found in sand dunes in humid areas. The Great Soil Group system is now superseded by the Australian Soil Classification.
PODZOLIC
There are several podzolic great soil groups, which are duplex or gradational soils, mostly with A2 horizons, and acid to neutral pH. The Great Soil Group system is now superseded by the Australian Soil Classification.
POLYHEDRAL
A type of structure with approximately equidimensional peds that have more than six faces and fit one into another in the soil profile.
PONDING
Accumulation of free water on the soil surface.
POOL
In nutrient cycling a pool is a defined form or location in which a nutrient element occurs within the ecosystem. For example, in the soil-plant N cycle, pools could include soil inorganic N, soil organic N and plant N.
PORES
That part of the soil not occupied by solid particles. Synonymous with voids. Soil pores are filled by air or water. The pore size distribution is the relative proportions of pores of different sizes in the soil.
POROSITY
The volume of soil pores (water + air filled) divided by the total soil volume.
POTENTIAL GRADIENT
The rate at which a potential varies with distance. For many quantities their flux is proportional to a potential gradient. For example plant water uptake from the soil is proportional to the water potential gradient.
PRECIPITATION
Precipitation has two different meanings in the context of soils. Precipitation refers to material falling to the earth's surface from the atmosphere, and in particular means the sum of rain and snow. Chemically, however, precipitation means the formation an insoluble substance by the chemical reaction of substances in solution.
PRESSURE POTENTIAL
The component of water potential that is caused by the hydrostatic pressure of overlying water. Pressure potential is zero in unsaturated soil. Below a water table pressure potential is positive and increases with depth. Pressure potential is sometimes lumped with matric potential. See also water potential.
PRIMARY PARTICLE
The individual mineral particles of which soil is composed, and which the soil can be separated into in the laboratory by dispersion techniques. Primary particles are those which comprise the clay, silt, sand and gravel fractions of the soil. Contrast with aggregate.
PRISMATIC
A type of soil structure where peds have a vertical axis that is much longer than the horizontal axis and have angular edges. Compare with columnar structure.
QUARTZ
A primary mineral, chemically silicon dioxide, which is abundant in all acid igneous rocks, as well as in many sedimentary and metamorphic rocks. It is resistant to weathering and is the main component of the sand fraction of most soils.
R HORIZON
Bedrock beneath the soil profile.
RED CLAY
A member of the grey, brown and red clays great soil group. Deep, uniform cracking clay soils widespread in the alluvial plains of inland Australia. The Great Soil Group system is now superseded by the Australian Soil Classification.
RED EARTH
A great soil group characterised by a red, uniform to gradational, weakly pedal profile. The Great Soil Group system is now superseded by the Australian Soil Classification.
RED-BROWN EARTH
A great soil group characterised by red-brown gradational to duplex profiles, sometimes with an A2 horizon, alkaline at depth with calcium carbonate concretions. The Great Soil Group system is now superseded by the Australian Soil Classification.
REGOLITH
The layers of unconsolidated material at the earth#39;s surface, comprising the solum and any underlying weathered rock.
RESIDUE
The organic remains of plants or animals.
RESPIRATION
The biochemical processes by which organisms obtain energy by oxidising sugars to carbon dioxide and water. In aerobic respiration molecular oxygen (O2) is used as the oxidising agent. In the absence of oxygen (eg in waterlogged soils), other oxidising agents can be used, including nitrate and ferric ions. This form of respiration is termed anaerobic respiration.
RHIZOSPHERE
The zone in the soil that is adjacent (eg within 1 mm) to the surface of a root.
RHYODACITE
A fine grained acid igneous rock.
RILL
A small erosion channel cut into the soil by a concentration of runoff. Contrast with gully. See also sheet erosion.
RUDOSOL
A soil order in the Australian Soil Classification. Rudosols are soils which have negligible pedologic organisation (ie negligible evidence of the effects of soil forming processes like horizon formation, colour differentiation or formation of peds). Very young soils might be Rudosols.
RUNOFF
Water that flows over the soil surface and away from a site. Contrast with runon.
RUNON
Water that flows over the soil surface and onto a site. Contrast with runoff.
SALINISATION
The process by which soil becomes saline. Saline soils are those containing high enough concentrations of soluble salts to adversely affect the growth of most plants. Primary salinisation is a natural soil forming process, but secondary salinisation is salinisation induced by human activity.
SALTATION
In wind or water erosion it is the transport of particles over the soil surface in a series of short jumps.
SAND
The International particle size classification scheme (used in Australia) defines sand as particles sizes between 2 mm and 0.02 mm in diameter. Coarse sand is from 2 to 0.2 mm, and fine sand is from 0.2 to 0.02 mm.
SAPROLITE
Soft weathered rock.
SATURATION
Soil is at saturation when all the pores are filled with water.
SELF-MULCHING
A surface soil condition where a loose mass of fine peds forms a natural mulch.
SESQUIOXIDES
A group of soil minerals comprising the oxides and hydroxides of iron and aluminium.
SHEET EROSION
A form of water erosion in which a thin uniform layer is removed from the soil surface. See also rill and gully.
SILICATES
Minerals containing silicon and oxygen as the main elemental constituents.
SILICEOUS SAND
A great soil group characterised by a sandy profile where there has been little profile development apart from the possible accumulation of surface organic matter. The Great Soil Group system is now superseded by the Australian Soil Classification.
SILT
In the International system (used in Australia), silt is defined as the particle size range between 0.002 mm and 0.02 mm in diameter.
SLAKING
The breaking down of aggregates to smaller aggregates due to internal stresses that accompany wetting. Contrast with dispersion.
SLICKENSIDES
Polished surfaces of B horizon peds that occurs in cracking clays as they shrink and swell during drying and wetting cycles.
SMECTITE
Group of 2:1 clay minerals characterised by high surface areas and cation exchange capacities, and variable interlayer spacing. Montmorillonite is a smectite commonly found in soils.
SODIC
Sodic soils are defined in Australia as those having an ESP (exchangeable sodium percentage) equal to or greater than 6%.
SODOSOL
A soil order in the Australian Soil Classification. Sodosols have a strong texture contrast between the A and B horizons, and the B horizons are sodic and not strongly acid.
SOIL COLOUR
See Munsell colour.
SOIL PROFILE
A vertical section through the soil.
SOIL REACTION
An old term meaning soil pH.
SOIL SOLUTION
Soil water and substances dissolved in it (ie solutes).
SOIL SURVEY
The systematic examination, description, classification, and mapping of soils in an area.
SOLODIC
A great soil group characterised by a duplex profile with a bleached A2 horizon and a sodic B horizon which is alkaline at depth. The Great Soil Group system is now superseded by the Australian Soil Classification.
SOLUM
(Plural: sola). The A and B horizons of the soil profile.
SOLUTE POTENTIAL
Also termed osmotic potential. That component of the soil water potential which is caused by the osmotic effect of solutes in soil water. The solute potential is always negative, becoming more so as the salt concentration increases. See also water potential.
SPECIFIC SURFACE AREA
Surface area per unit volume or mass
STRENGTH
Soil strength is a measure of the resistance of soil to externally applied forces.
STRIP CROPPING
A strategy to reduce water or wind erosion in which different crops are grown in strips across the slope, in such a way that no adjacent strips are bare at any one time.
STRUCTURE
Soil structure is the three dimensional arrangement of primary particles in the soil profile. Primary particles are often bonded together to form peds or natural aggregates. In the field, soil structure is characterised by describing the shape (type), size and distinctness (grade) of the peds.
SUBANGULAR BLOCKY
Subangular blocky peds are square or block like with rounded edges.
SURFACE SEALING
Sealing of the soil surface during rain, resulting from breakdown of aggregates. May lead to the formation of a crust when the soil dries.
SURFACE TENSION
The force per unit length necessary to pull apart a liquid surface.
SUSPENSION
A fluid containing particles which are small enough to settle out only slowly, but are not dissolved.
TENOSOL
A soil order in the Australian Soil Classification. Tenosols include a wide range of soils which have only weak pedologic organisation (ie negligible evidence of the effects of soil forming processes like horizon formation, colour differentiation or formation of peds) apart from the formation of A horizons. Contrast with Rudosols.
TENSIOMETER
A suction device that is used to measure the soil matric potential in the field.
TETRAHEDRON
A volume with four sides and four vertices. In soil clay minerals a silicon (+4) cation is often surrounded by 6 oxygen anions situated at the vertices of a tetrahedron. Linked tetrahedrons form the tetrahedral layers in silicate clay mineral crystals.
TEXTURE
Soil texture is the relative proportions of the sand, silt and clay fractions in the soil. These fractions can be determined quantitatively in the laboratory, and the soil texture triangle is then used to assign a texture grade (eg sandy loam). In the field the ribbon test is used to determine the texture grade. Field texture is not identical to that determined in the laboratory, because factors apart from particle size distribution can influence the field method.
TILLAGE
Any process which disturbs the upper part of the soil profile, mostly for the purpose of aiding crop production. Tillage is used to kill weeds, incorporate plant residues, decrease soil strength, decrease aggregate size, increase porosity, level the soil surface, etc.
TOPOSEQUENCE
A series of soils for which all the factors of soil formation apart from topography are approximately constant. For example a sequence of soils formed from the same parent material down a hill slope usually forms a toposequence. Toposequence can be taken as synonymous with the older term catena.
TRACE ELEMENTS
Elements present in small quantities. Sometimes used synonymously with micronutrients.
TRANSPIRATION
Loss of water as vapour to the atmosphere that has been taken up from the soil by plants. Contrast with evaporation and evapotranspiration.
TURGOR PRESSURE
Pressure exerted on the cell walls by water contained within the cell. Turgor pressure prevents plant tissues from wilting and drooping.
UNIFORM
A uniform soil is one which has a uniform texture profile, which means that the texture remains approximately the same at all depths.
UNIVERSAL SOIL LOSS EQUATION
An equation developed in the USA to predict soil loss by water erosion in that country. Modifications have been made to allow the USLE to be used successfully in parts of Australia.
UREA
The dominant form of N in urine and a widely used N fertiliser.
USLE
See universal soil loss equation.
VALUE
In the measurement of soil colour, the value indicates how light or pale the colour is (ie white has a high colour value, dark grey has a low value). See also Munsell colour.
VARIABLE CHARGE
Electrical charge present on soil colloids that varies with the soil chemical environment, particularly with pH. Contrast with permanent charge.
VERTOSOLS
A soil order in the Australian Soil Classification. Vertosols are clay soils which exhibit shrink-swell characteristics, crack strongly when dry and have lenticular peds and/or slickensides at depth.
VOID RATIO
The volume of soil pores divided by the volume of solid matter.
VOLATILISE
Transform from liquid or solid state to gaseous state. See also ammonia volatilisation.
VOLUMETRIC WATER CONTENT
Volume of soil water per unit bulk volume of soil.
WATER HOLDING CAPACITY
The amount of water held by soil at field capacity.
WATER POTENTIAL
The soil water potential is the potential energy per unit quantity of soil water. Its value in understanding the behaviour of soil and plant water is based on the fact that water can only move spontaneously from a place of high potential to one of low potential, and that the rate of water movement is proportional to the potential gradient (See also hydraulic potential). Soil water potential can be split into a number of component potentials including matric potential, pressure potential, gravitational potential and solute potential.
WATER TABLE
The upper surface of free water in the soil. Below the water table the soil is saturated.
WATERLOGGED
Saturated with water.
WEATHERING
Those chemical and physical processes which by which minerals are physically altered and chemically decomposed near the earth's surface by air and water.
WILTING POINT
The water content or soil water tension at which plants lose turgor and wilt. See also permanent wilting point.
YELLOW EARTH
A great soil group characterised by a yellow to yellow-brown, uniform to gradational, weakly pedal profile. The Great Soil Group system is now superseded by the Australian Soil Classification.